Vaccines
 Vaccine Impact from Evidence to Policy
Vaccine Impact from Evidence to Policy💉 Transforming Vaccine Science into Policy and Practice
From Real-World Evidence to Global Decisions and Community Impact
📜 Background
Vaccination is one of the most powerful public health interventions. Real-world effectiveness, population impact, and implementation strategies are critical to inform policy decisions, particularly during pandemics and emerging outbreaks.
During the COVID-19 pandemic, rapid and robust evidence on vaccine effectiveness (VE) was urgently needed to guide global immunization strategies, emergency authorizations, and public trust. At the same time, implementation in vulnerabilized populations required context-specific, community-driven approaches.
Our work spans the full translational spectrum, from methodological innovation and large-scale effectiveness studies to direct implementation in vulnerabilized communities. These interventions were developed horizontally and co-designed with communities and civil society, engaging stakeholders across multiple levels, including local leaders, politicians, municipal authorities, ministries of health, and academia. This approach was grounded in best-practice implementation science frameworks, incorporating principles of patient and public involvement and engagement (PPIE) to ensure acceptability, equity, and sustainability of vaccination strategies.
🔬 Our Contribution
- Vaccine Effectiveness (VE) Science: Led and contributed to large-scale observational studies evaluating COVID-19 vaccines using rigorous designs (e.g., test-negative case-control; target trial emulations).
- Methodological Innovation: Advanced analytical approaches for VE estimation under real-world conditions, including bias control and rapid evidence generation during epidemics.
- Global Collaboration (VEBRA): Co-led the VEBRA consortium, integrating data across Brazil and international partners (Fiocruz, UFMS, IR Sant Pau, Yale, Stanford, Emory, Johns Hopkins, University of Florida).
- Implementation Science: Designed and evaluated large-scale vaccination strategies in vulnerable settings, including the Vacina Maré initiative in Rio de Janeiro.
- Policy Engagement: Contributed evidence used in national and international decision-making, including WHO-linked processes.
X0002-6#) (2022, *left*) and [The Lancet Infections Diseases](https://www.thelancet.com/journals/laninf/issue/vol26no1/PIIS1473-3099(25)X0013-X) (2026, *right*)](/media/cover_vaccine_lancet_hu10210480853062511692.webp)
🌍 Impact
- Global Policy and Emergency Use:
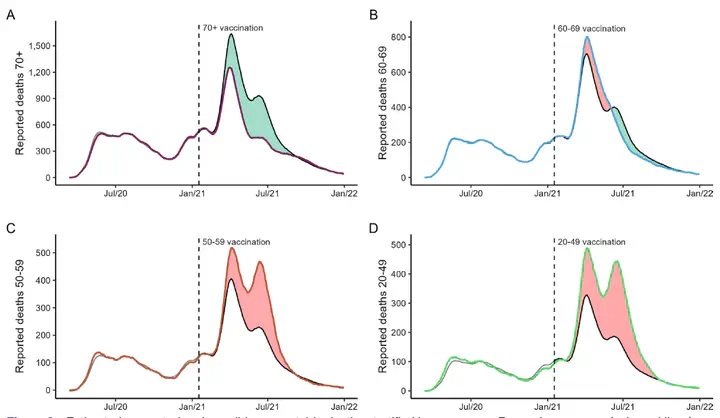
- Our landmark studies in The Lancet Regional Health Americas and The BMJ on CoronaVac were among the first large-scale real-world evaluations of an inactivated COVID-19 vaccine. The BMJ manuscript had massive dissemination both as a pre-print and when published. We presented it at the WHO SAGE Committee and it was one of the main pillars supporting the vaccine’s emergency authorization, particularly for having data on those aged ≥70 years who were not included in the phase-3 randomized trials. By Jan 2022, CoronaVac (Sinovac) was the most used vaccine in the world.
- Overall evidence from VEBRA-COVID contributed to emergency use decisions and global policy discussions, particularly in low- and middle-income countries.
- Findings were cited in WHO and national immunization guidelines, patents, Wikipedia and broadly discussed in the academic and lay media, influencing vaccine deployment strategies worldwide.

Scientific Breakthrough in Real-World VE:
- Established robust evidence on vaccine performance against Gamma and Omicron variants, including booster strategies.
- Contributed to defining methodological standards for VE studies, widely adopted during the pandemic.
Implementation at Scale: #VacinaMaré

- Co-led a community-based mass vaccination campaign in one of Brazil’s largest favelas.
- The first phase of the mass vaccination campaign was performed in the 16 slums of Maré. It occurred between July 29 and August 1, 2021, which immunized more than 36,000 residents over the age of 18 in just four days.
- Achieved near-universal vaccine coverage, demonstrating feasibility of equitable vaccine delivery.
- Generated direct reductions in cases and transmission, while strengthening trust between communities and health systems.
- Became a model for implementation science in vulnerabilized settings, integrating civil society, local leaders, and public health authorities.
- Covered in the New York Times
Impact on Global Health Institutions:
- Evidence contributed to WHO SAGE deliberations and broader vaccine policy frameworks.
- Influenced decisions across ministries of health, PAHO, WB, and international agencies.
- Work cited in policy documents, media, and global health reports, amplifying its reach beyond academia.
Next-Generation Vaccines - Dengue:
- First real-world evaluation of TAK-003 dengue vaccine effectiveness (Lancet Infectious Diseases, 2026).
- Researcher in the community-based mass vaccination campaign in the city of Dourados, Brazil (~250,000 inhabitants), for TAK-003, still on evaluation.
- Provided critical evidence for national immunization programs, particularly in the context of prior safety concerns with dengue vaccines.
- Supports safe and evidence-based deployment in endemic regions.
🤝 Global Collaborative Network
This work was conducted through a multinational research network, integrating academic institutions, public health agencies, and local stakeholders, always with active collaboration with PAHO, Brazilian Ministry of Health, State and Municipal Health Secretaries.
For instance, we are leading partners on the VEBRA Research Network on Vaccines.

🧠 Key Insight
High-quality real-world evidence, when combined with strong partnerships and community engagement, can directly shape global health policy and deliver measurable benefits to populations.


 This work was supported in part by the
Pan American Health Organization (PAHO),
the Bill & Melinda Gates Foundation, CNPq,
ITpS, CDC and many other partners and stakeholders.
This work was supported in part by the
Pan American Health Organization (PAHO),
the Bill & Melinda Gates Foundation, CNPq,
ITpS, CDC and many other partners and stakeholders.📚 Selected References
Ranzani OT, Lazar Neto F, Mareto LK, et al. (2026). Effectiveness of the TAK-003 dengue vaccine in adolescents during the 2024 outbreak in São Paulo, Brazil: a test-negative, case-control study. Lancet Infect Dis. 26(1):91-100. doi: 10.1016/S1473-3099(25)00382-2.
Batista-da-Silva AA, Bastos LSL, …, Ranzani OT. (2024) #VacinaMare campaign: addressing vaccine inequity in socially vulnerabilised communities. Lancet Reg Health Am. 36:100827. doi: 10.1016/j.lana.2024.100827.
Ranzani OT, Hitchings MDT, de Melo RL, et al. (2022). Effectiveness of an inactivated Covid-19 vaccine with homologous and heterologous boosters against Omicron in Brazil. Nat Commun. 13(1):5536. doi: 10.1038/s41467-022-33169-0.
Hitchings MDT, Lewnard JA, …, Ranzani OT, …, Cummings DAT. (2022). Use of Recently Vaccinated Individuals to Detect Bias in Test-Negative Case-Control Studies of COVID-19 Vaccine Effectiveness. Epidemiology. 33(4):450-456. doi: 10.1097/EDE.0000000000001484.
Ranzani OT*, Hitchings MDT*, Dorion M, et al. (2021). Effectiveness of the CoronaVac vaccine in older adults during a gamma variant associated epidemic of covid-19 in Brazil: test negative case-control study. BMJ. 374:n2015. doi: 10.1136/bmj.n2015.